What is Salvia Miltiorrhiza Extract?
Salvia miltiorrhiza Extract Powder is a standardised traditional Chinese medicine raw material that is extracted, separated, purified, and dried from the dried roots and rhizomes of Salvia miltiorrhiza Bge. Using modern phytochemical extraction techniques.
There is a fundamental difference between Salvia miltiorrhiza Extract Powder and Danshen powder, which is directly crushed from the Danshen medicinal herbs. Salvia miltiorrhiza Extract Powder enriches key medicinal substances in Danshen through scientific processes, such as water-soluble Danshenphenolic acid components represented by Danshensu B and fat-soluble Danshenketone components represented by Danshensu IIA.

COA
|
General Information |
|
|
Name |
Salvia miltiorrhiza Extract Powder |
|
Part used |
Root |
|
Item |
Specification |
|
Appearance |
Brownish-red Powder |
|
Flavor & Odor |
Characteristic |
|
Particle Size |
95% pass 80 mesh |
|
Physical |
|
|
Loss on Drying |
≤5.0% |
|
Bulk Density |
40g-60g/100ml |
|
Chemical |
|
|
Pb |
≤3mg/kg |
|
As |
≤1mg/kg |
|
Hg |
≤0.1mg/kg |
|
Cd |
≤1mg/kg |
|
Microbial |
|
|
Total microbacterial count |
≤1000cfu/g |
|
Yeast & Mold |
≤100cfu/g |
|
E.Coli |
Negative |
|
Staphylococcus aureus |
Negative |
|
Salmonella |
Negative |
|
Enterobacteriaceaes |
Negative |
Chromatogram
Unlike cosmetics or dietary supplements that often use a single solvent (such as ethanol or water) for simple extraction, we have developed a refined extraction process aimed at comprehensively and efficiently obtaining water-soluble and lipophilic dual active ingredients from Salvia miltiorrhiza Extract, tailored to the characteristics of traditional Chinese medicine applications.
- Fresh fruits are washed.
- Filter
- Remove impurities
- Drying: to prevent high-temperature damage to thermosensitive active ingredients, especially volatile oils.
- Crushing: (such as 80 mesh) to increase the contact area with the solvent and improve extraction efficiency.
- Core extraction
- Solid-liquid separation
- Purification
- Concentrate
- Drying
- Smash
- Packaging
- Finished product
Key Quality Control Indicators and Testing Methods
Determination of indicator component content:
- Salvianolic acid B: Quantitative detection was performed using high-performance liquid chromatography (HPLC).
Reference pharmacopoeia and related research for chromatographic conditions: octadecylsilane bonded silica gel was used as the filler (C18 column); The mobile phase is usually a gradient elution of acetonitrile phosphate aqueous solution; The detection wavelength is set at around 280 nm.
We ensure that the content of salvianolic acid B meets the specifications agreed upon by both parties, and that the linear range, precision, and recovery rate have been rigorously validated by methodology.
Structural Formula:
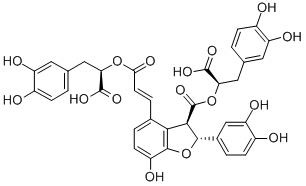
- Tanshinone IIA: also determined by the HPLC method. The mobile phase is usually a methanol-water or acetonitrile-water system; The detection wavelength is set at 270 nm. We precisely control the content of tanshinone IIA to ensure it is within the specified range.
Structural Formula:
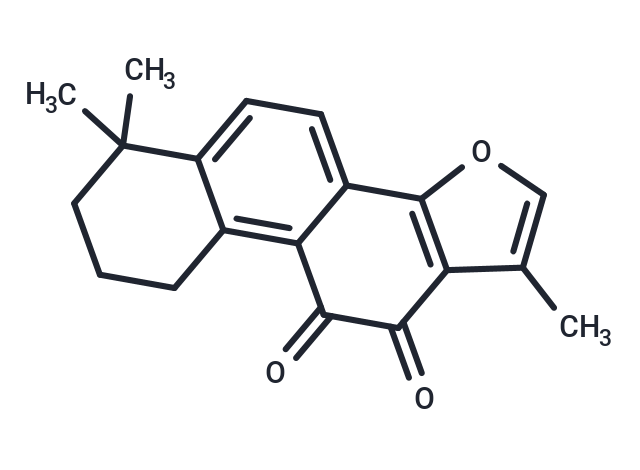
Is Salvia Miltiorrhiza Extract Effective?
The widespread application of Salvia miltiorrhiza Extract is rooted in its solid pharmacological foundation.
cardiovascular and cerebrovascular system
The salvianolic acid components in Salvia miltiorrhiza Extract, especially salvianolic acid B, can protect vascular endothelial cells and maintain vascular elasticity by scavenging free radicals and inhibiting low-density lipoprotein (LDL) oxidation. Meanwhile, tanshinone components can inhibit platelet aggregation and adhesion, reduce blood viscosity, and improve microcirculation.
Regulating effect on the central nervous system
Salvia miltiorrhiza Extract can promote the release of brain-derived neurotrophic factor (BDNF) by activating the mTOR signalling pathway in the brain, thereby mediating rapid antidepressant effects. In addition, Salvia miltiorrhiza Extract can also regulate the central nervous system neurotransmitter system, exerting sedative and anti-anxiety effects.

Certificates

Security statement:
Our company guarantees that the goods sent are safe, non-flammable, and non-explosive, and consistent with the declaration. Under normal transportation conditions, all losses caused by the products sent by our company shall be borne by our company.
Hot Tags: Salvia Miltiorrhiza Extract manufacturers, suppliers, factory, wholesale, best, price, buy, for sale, bulk

FAQs
Q: What are the core differences between APPCHEM's Salvia Miltiorhiza Extract and ordinary Danshen powder on the market?
A: The core difference lies in standardisation and purity. Ordinary Danshen powder is a physical pulverisation of medicinal materials, with unstable active ingredient content and a large amount of ineffective plant fibres. Our Salvia Militiorhiza Extract is a product that has been scientifically extracted, separated, and purified. The active ingredients are highly enriched, and each batch has clear indicators of active ingredient content (such as salvianolic acid B and tanshinone IIA). The quality is stable and uniform, with better solubility and higher bioavailability, fully meeting the requirements of modern pharmaceuticals.
Q: What are the recommended storage conditions and shelf life for the product?
A: It is recommended to seal and store the product in a cool, dry, and dark environment, avoiding high temperatures and humidity. The shelf life of the product is 24 months under recommended storage conditions
Q: How to ensure the stability of product quality between different production batches?
A: We ensure consistency between batches through a triple guarantee system: 1) strict raw material control: using high-quality Salvia Militorhiza from the local production area and conducting incoming testing on each batch of raw materials; 2) Standardized Production Process (SOP): From feeding to packaging, every step strictly follows validated standard operating procedures; 3) Comprehensive quality testing: Each batch of finished products must undergo a comprehensive testing including TLC identification, HPLC fingerprint, and multi index component quantification to ensure that their chemical spectrum and key component content are highly consistent with the standard.
Q: As a raw material, what is the recommended dosage for its use in end products?
A: As a B2B raw material supplier, we do not directly recommend the final dosage of end products. The dosage design needs to be determined by your formula engineer based on the product type (drugs, health food), traditional Chinese medicine formula theory (such as the principle of compatibility between Jun Chen Zuo), target population, and relevant regulatory requirements. As a reference, the standard dosage for Danshen decoction pieces in the Chinese Pharmacopoeia is 10-15 grams per day
Reference
[1]Study on the Mechanism of Salvia Miltiorrhiza in the Treatment of Prostatic Hyperplasia Based on Online Pharmacology. Shaoyang Hou et al. [2025-05-07]
[2]Danshensu Interventions Mediate Rapid Antidepressant Effects by Activating the Mammalian Target of Rapamycin Signalling and Brain-Derived Neurotrophic Factor Release. Han-Wen Chuang et al. [2024]
[3]Traditional Chinese medicine of Salvia miltiorrhiza Bunge: a review of phytochemistry, pharmacology and pharmacokinetics. Ebuka-Olisaemeka Nwafor. [2021-01-01]
[4]Phytochemical Constituents and Pharmacological Effects of Dangshen (Codonopsis pilosula), an Important Traditional Chinese Medicine. Li Yani et al. [2025-12-02]
[5]Xihong Wang, S. Morris-Natschke et al. "New developments in the chemistry and biology of the bioactive constituents of tanshen." Medicinal Research Reviews (2007). [2007-01-01]







