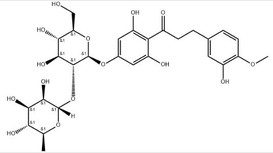
What Is This High-Intensity Sweetener from Citrus?
Neohesperidin Dihydrochalcone (NHDC) is a derivative of flavonoid compounds originating from citrus fruits, particularly bitter orange (Citrus aurantium). Produced through catalytic hydrogenation of naturally occurring neohesperidin in citrus fruits, it serves as a high-intensity, calorie-free sweetener and flavour modifier.
The market demand for reduced-sugar products continues to grow. However, many artificial sweeteners present challenges such as poor thermal stability, unpleasant aftertastes, or consumer concerns regarding "chemically synthesised" labels. These issues pose difficulties for B2B clients in formulation development, clean label claims, and consumer acceptance.

To address this, we introduce this 98% high-purity Neohesperidin Dihydrochalcone (NHDC), rigorously quantified via HPLC. It is not merely an ingredient, but a comprehensive natural sugar-reduction solution for food and beverage R&D engineers, pharmaceutical formulation specialists, procurement managers, and quality decision-makers. Strictly adhering to international mainstream food safety standards, its core value lies in: natural origin, HPLC precision quantification, multifunctionality, and full traceability. We are committed to presenting a complete quality evidence chain from citrus to finished product through this detailed product dossier, serving as a reliable component in your product innovation journey.
PS: This page focuses on the product Technical Data Sheet.
For the product application information, please click High-concentration Sweetener Ingredient NHDC Powder.
For Integrated Supply Chain & Solutions, please click NHDC Powder ISO-Certified Original Factory.
What Are Its Properties And Compliance Criteria?
Physical and Chemical Properties
| Common Name | Neohesperidin Dihydrochalcone, NHDC |
| Other Name | Neohesperidin DC, E 959 |
| CAS Number | 20702-77-6 (Unique Chemical Substance Identifier) |
| Molecular Formula |
C₂₈H₃₆O₁₅
|
| Molecular Weight | 612.58 g/mol |
| EINECS | 200-488-7 |
| Melting point | 156-158 °C(lit.) |
| Boiling point | 927.1±65.0 °C(Predicted) |
| Density | 1.61±0.1 g/cm3(Predicted) |
| Acidity constant (pKa) | 6.85±0.40(Predicted) |
| LogP | 0.67 at 20℃ |
| FEMA | 3811 |
| Merck | 6452 |
| Appearance and Characteristics | Off-white to white powder, free from lumps and visible foreign matter |
| Solubility | Soluble in water (approximately 2.04 g/L), methanol, ethanol, DMSO and other solvents. Its water solubility may be improved through blending with other sweeteners. |
| Stability | Possesses excellent thermal stability and acid-base stability, remaining stable at temperatures up to 160°C and across a broad pH range. Suitable for products requiring high-temperature processing or long-term storage. Recommended for storage in a cool (<25°C), dry, dark environment in sealed containers, with a shelf life of 24-36 months. |
Safety and Compliance Criteria
This product's safety standards comprehensively reference regulatory requirements from major markets, including China, the European Union, and the United States. Both internal and third-party laboratories have validated all data.
| Heavy Metal Limits |
Complies with stringent restrictions outlined in the 2020 edition of the Chinese Pharmacopoeia and the EU Cosmetics Regulation (EC) No 1223/2009: Lead (Pb) ≤ 1 mg/kg Arsenic (As) ≤ 1 mg/kg Cadmium (Cd) ≤ 1 mg/kg Mercury (Hg) ≤ 0.1 mg/kg |
| Pesticide Residues | Screening employs GC-MS/MS and LC-MS/MS methods to ensure no detection or levels below the limit of quantification for over 200 common pesticides. |
| Microbial Control |
Total Aerobic Bacteria ≤ 1,000 CFU/g Total mould and yeast count ≤ 100 CFU/g Not detectable: Escherichia coli, Salmonella spp., Staphylococcus aureus |
| Non-GMO and Non-Irradiated Declaration | The raw materials for this product are sourced from non-genetically modified citrus resources. Throughout the entire production and processing stages, no genetically modified techniques or irradiation sterilisation treatments have been employed, and a written declaration is provided. The product complies with Kosher and Halal certification requirements. |
How to Interpret Product Specifications?
As a high-intensity sweetener, the purity of Neohesperidin Dihydrochalcone (NHDC) directly impacts its sweetness profile, application efficacy and cost-effectiveness. Standard Specification: HPLC ≥ 98% Neohesperidin Dihydrochalcone (NHDC). This represents the internationally recognised high-purity standard within the food and pharmaceutical industries, suitable for the vast majority of commercial applications.
| Specification | Typical Content | Cost Implications | Efficacy & Application Differences |
| Standard Grade | 90% - 95% | Economical | Suitable for cost-sensitive foundational products with broad efficacy targeting; formulation loading capacity must be adequately reserved. |
| High-Purity Grade | 96% - 98% | Optimal cost-effectiveness | Active ingredients are clearly defined, with precise calculation of addition levels. Suitable for sectors demanding strict purity, safety, and sensory quality standards, including food and beverages, health supplements, and pharmaceuticals. |
Key Considerations: Selecting products of different specifications primarily involves balancing cost, application areas, and regulatory requirements. Regardless of the specification you choose, you will receive our consistent assurance of reliability in raw material sourcing, safety testing, and quality control systems.
What Does the COA Include?
We undertake to provide a Certificate of Analysis (COA) compliant with international standards with every consignment. The development and validation of our testing methodologies incorporate authoritative standards such as the United States Pharmacopoeia, European Pharmacopoeia, and Chinese Pharmacopoeia to ensure global data compliance. Furthermore, every test result on the COA is traceable to its original records and supports audit verification by clients or third parties.
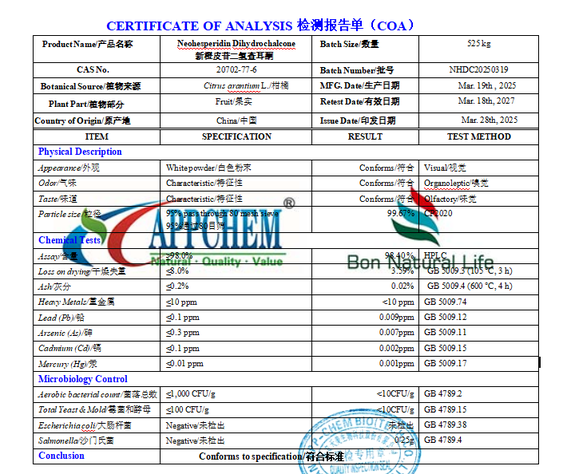
Important Notice: The COA information provided on this page is for illustrative purposes only, intended to demonstrate our quality control dimensions and capabilities. For the specific COA documentation of actual products (including precise batch numbers, HPLC test data, and chromatograms), please refer to the final version issued by our business specialists.
What's the Product Process Flow?
We employ our proprietary green preparation process to efficiently extract and purify high-purity neohesperidin dihydrochalcone from natural citrus raw materials. The diagram below illustrates the key process flow from raw material to finished product.

Further Information: The flowchart above outlines the primary stages of the process. For more detailed production information regarding specific process parameters, hydrogenation catalyst selection, capacity specifications, and bespoke manufacturing, please contact our business specialists. We shall provide further technical consultation tailored to your specific requirements.
How Does the End-to-End Quality Management Ecosystem Work?
Our quality management system constitutes a closed-loop ecosystem spanning the entire lifecycle from cultivation and production through testing to release. Its core principles are "prevention-oriented, fully controllable, and data traceable".
Source Control (Upstream)
- Origin Traceability: Collaborate with audited contract citrus cultivation bases to ensure transparent sourcing and consistent raw material quality.
- By-product Utilisation: Primarily employs citrus processing industry by-products (such as peel residue) as starting materials, aligning with circular economy and sustainable development principles.
- Dual Verification for Raw Material Entry: Each batch must be accompanied by a certificate of origin. Upon arrival, materials undergo preliminary QC screening (moisture content, neohesperidin screening). Only compliant batches enter the holding area for comprehensive pesticide residue and heavy metal screening, ensuring only premium raw materials enter production.
Production Process Control (Midstream)
- HACCP-based Critical Point Management: Multiple Critical Control Points are established throughout the production line. For instance, online monitoring is implemented during hydrogenation (CCP1), crystallisation purification (CCP2), and drying (CCP3) processes to ensure operations remain within pre-validated "design space" parameters.
- Batch Clarity Management: Rigorous implementation of area clearance protocols, material identification systems, and electronic batch record systems eliminates any potential for confusion or cross-contamination.
Laboratory Quality Assurance (Downstream)
- Release Testing and Stability Studies: Each batch of finished product must pass comprehensive testing covering all physicochemical and safety parameters. We utilise advanced analytical instruments, including HPLC, GC, and UV, to assess multiple quality parameters such as NHDC content, related substances, loss on drying, heavy metals, and microbiological counts.
- Stability Testing: Conduct long-term and accelerated stability studies in accordance with the principles of internal quality control for enterprise standards to scientifically determine shelf life, ensuring consistent product quality throughout the declared period.
What Certifications and Support Documents Are Available?
Our transparent quality commitment and R&D capabilities are underpinned by the following authoritative certifications, patented technologies, and supporting documentation.

System Certification Certificates:
- ISO 22000 Food Safety System Certification
- ISO 9001 Quality Management System Certification
Key Supporting Documentation:
- FDA, KOSHER, HALAL, SC...
- Full-project Quality Inspection Reports (batch-specific)
- Material Safety Data Sheet (MSDS)
- Factory Audit Report Summary
We fully recognise the importance of reviewing original documentation during supplier selection. Should you require access to originals or certified copies of the aforementioned certificates, patent documentation, or batch-specific supporting files for formal supplier evaluation or reference purposes, please contact our dedicated manager. We shall provide full cooperation in supplying the required materials to support your decision-making process.
What Applications and Custom Formulation Support Are Offered?
Our transparent quality commitment and R&D capabilities are underpinned by the following authoritative certifications, patented technologies and supporting documentation.
With its well-defined mechanism of action, high-purity NHDC serves as an ideal raw material for product innovation across multiple industries. Core Functional Applications:
- High-Intensity Sweetener
- Bitterness Masker
- Flavour Enhancer
- Synergistic Effects
Recommended Formulation Solutions:
- Reduced-Sugar Beverage Solution: NHDC + Stevioside/Erythritol.
- Pharmaceutical Taste Optimisation Solution: NHDC + Aspartame/Acesulfame K.
- High-Protein Nutrition Bar Solution: NHDC + Maltitol/Isomalt.
Customised Solutions: Our dedicated application R&D team provides tailored technical support-from formulation optimisation and stability studies to sample production-for your specific product concepts (e.g., sugar-free vitamin gummies for children, diabetes-friendly protein drinks, plant-based functional beverages).
Contact Us: For specific batch COAs, TDS, process details, samples, or to discuss bespoke requirements, please contact our technical sales team. Complimentary samples are available for testing; contact us before ordering to arrange dispatch.
Reference
1. Horowitz, R. M., et al. (1963). Neohesperidin dihydrochalcone: A new sweetening agent. Journal of Agricultural and Food Chemistry, 11(5), 345-348. doi:10.1021/jf60132a013
2. Waalkens-Berendsen, D. H., et al. (2004). Embryotoxicity and teratogenicity study with neohesperidin dihydrochalcone in rats. Regulatory Toxicology and Pharmacology, 40(1), 74-79. doi:10.1016/j.yrtph.2003.10.005
3. Gu, L., et al. (2004). Antioxidant activity of neohesperidin dihydrochalcone from citrus extract. Food Chemistry, 86(4), 557-562. doi:10.1016/j.foodchem.2003.10.009
4. Khan, N., et al. (2019). Neohesperidin dihydrochalcone (CAS 20702-77-6) inhibits oxidative stress and inflammation in human cells. Journal of Functional Foods, 59, 103012. doi:10.1016/j.jff.2019.103012
5. Matsumoto, K., et al. (2017). Neohesperidin dihydrochalcone protects intestinal epithelial cells from oxidative damage via activating the Nrf2/ARE signalling pathway. Biochemical and Biophysical Research Communications, 483(3), 1024-1030. doi:10.1016/j.bbrc.2016.12.084
6. Prior, R. L., et al. (2001). Sweetness and antioxidant activity of citrus flavonoids, including neohesperidin dihydrochalcone. Journal of Agricultural and Food Chemistry, 49(10), 4784-4790. doi:10.1021/jf0104874