What Is the Pomegranate Extract Punicalagin?
Pomegranate Extract Punicalagin is a class of highly active natural polyphenolic compounds extracted from pomegranate peel (Punica granatum L.). Punicalagin belongs to the ellagitannin group and is one of the most abundant and bioactive components within pomegranate polyphenols. Within the global plant extract market, punicalagin-rich pomegranate extract finds extensive application in dietary supplements, functional foods, botanical antioxidant formulations, and natural health product development.
For B2B purchasers, selecting pomegranate extract punicalagin raw materials requires consideration not only of active ingredient content but also of supply source stability, batch consistency, production capacity scale, and supply chain reliability. In international raw material trade, stable inventory and sustained supply capability often hold greater value than single-batch pricing. Consequently, a supply system offering Pomegranate Extract Punicalagin In Stock enables rapid delivery when required and supports long-term bulk procurement needs.
PS: This page focuses on the Integrated Supply Chain & Solutions.
For the product application information, please click Whitening Cosmetics Ingredient Pomegranate Extract Punicalagin.
For the product Technical Data Sheet, please click Patent Ingredient Punicalagin HPLC40% 65995-63-3.
What Are the Core Challenges and Scientific Responses in Punicalagin Industrialisation?
As a high-value characteristic component extracted from pomegranate peel, punicalagin faces several critical industrial-scale conversion bottlenecks in its transition from laboratory research to large-scale commercial application.
Challenge One: The Complex Relationship Between Macromolecular Structure, Water Solubility, and Stability
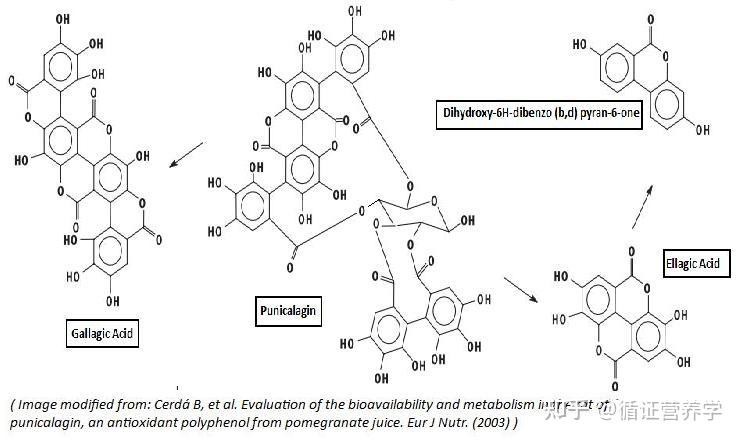
Punicalagin ranks among the largest known natural polyphenolic compounds by molecular weight (1084.72 Da). Its structure comprises gallic acid dimers (ellagic acid and gallic acid) linked to glucose via ester bonds. This macromolecular characteristic introduces three critical industrialisation issues:
- Dual water solubility characteristics: Unlike ellagic acid, which is slightly soluble in water, punicalagin itself exhibits good water solubility and readily enters the aqueous phase of pomegranate juice. However, its dissolution behaviour in high-purity form differs significantly from that in crude extracts. Industry practice indicates batch-to-batch variations in dissolution rates and equilibrium solubility across different purity grades of punicalagin, posing challenges for precise formulation in downstream applications.
- Environmental dependence of chemical stability: Multiple ester bonds within the punicalagin molecule render it sensitive to pH, temperature, and light exposure. Under alkaline conditions, hydrolysis readily occurs, releasing small-molecule products such as ellagic acid; prolonged high-temperature storage may also induce structural rearrangement or degradation. This implies that unstable production processes or storage conditions can cause drift in the product's active ingredient profile, leading to inconsistencies where "declared content meets standards but efficacy performance varies".
For B2B clients, this signifies that when procuring Punicalagin, the supplier's production process stability, batch consistency of purity specifications, and deep understanding of the product's physicochemical properties are more critical decision factors than price alone.
Our Scientific Response and Product Solutions:
As a source manufacturer, we have established a comprehensive technical control system spanning the entire chain from raw materials to finished products:
- Multi-specification System and Physical Property Stability Control: We offer Punicalagin products in multiple specifications (10%, 25%, 40%, etc.), establishing independent solubility curves and stability data archives for each specification. This ensures that downstream customers need not repeatedly adjust process parameters during formulation development due to fluctuations in physical properties between batches.
- Stability Data Support: We furnish accelerated stability testing (40°C/75% RH) and long-term stability data, specifying expected shelf life under varying storage conditions (standard conditions: 2 years). This enables clients to establish scientifically grounded inventory management protocols.
Challenge Two: Complexity of Metabolic Pathways and Scientific Design for Efficacy Validation
The metabolic pathways of punicalagin within the gastrointestinal tract are exceptionally intricate, presenting profound challenges for efficacy validation and product claims:
- Multi-stage Hydrolysis and Conversion Pathways: All ellagitannins (including punicalagin) undergo hydrolysis in the small intestine to yield ellagic acid. Unabsorbed punicalagin and ellagic acid entering the colon undergo further metabolism by gut microbiota through a series of decarboxylation reactions, ultimately yielding active metabolites such as urolithins A and B. This implies that punicalagin's biological activity in vivo is largely mediated by its urolithin metabolites rather than the parent molecule itself.
- Significant inter-individual metabolic variation: Research indicates that not all individuals can produce the final urolithin metabolites. Based on the types of urolithins detected in urine 24 hours post-ingestion, individuals can be categorized into three metabolic phenotypes: UM-A (producing only urolithin A), UM-B (producing urolithins A, B, and isourolithin A), and UM-0. This variation correlates positively with the abundance of Gordonibacter in the gut.
- Bioavailability limitations: Classified as a Category IV drug (low solubility, low permeability) under the bioavailability classification system, punicalagin exhibits an oral absorption rate of merely 3-6%, with the majority metabolized by colonic microbiota. In human studies, serum metabolite concentrations following 318mg punicalagin intake were detected at ng/mL levels.

For B2B clients, this implies: Verifying punicalagin's efficacy cannot rely on linear thinking that equates "in vitro activity with in vivo efficacy". A commercially viable punicalagin project requires end-to-end validation across the entire chain: "raw material composition profile → in vivo metabolic pathways → target mechanism of action → consumer-perceived efficacy". This must also account for metabolic phenotype distribution within the target population.
Our scientific response and product solutions:
- Batch consistency and traceability: We guarantee high inter-batch consistency in the raw material's composition profile, impurity profile, and key activity indicators. This forms the foundation for generating reproducible data in any subsequent in vivo or in vitro efficacy studies.
- Literature Support for Efficacy Studies: We provide clients with foundational research literature on Punicalagin and its metabolite punicalin, including data on antioxidant, anti-inflammatory, cardiovascular protective, anti-cancer, antibacterial, and antiviral activities. This supports clients in building scientific claims.
- Technology Consultancy: Addressing bioavailability challenges, we recommend enhancement strategies such as micronization, nano-encapsulation, and cyclodextrin inclusion complexes. We also support clients in conducting pilot-scale validation trials.
What Is the Production Capacity Planning and Bulk Supply Availability?
For international buyers, the availability of stable inventory directly impacts cooperation efficiency. The supply capability of Pomegranate Extract Punicalagin In Stock is underpinned by rational production capacity planning and raw material reserve strategies. As a seasonal fruit, pomegranate peel raw materials are typically sourced during concentrated harvest periods. Consequently, industrial production involves large-scale procurement during harvest seasons, with extended usage cycles achieved through drying or cold storage. This stockpiling strategy ensures year-round production continuity, mitigating the impact of seasonal fluctuations on supply.
At the production level, bulk orders are typically processed in batches. Even tonnage-scale orders are not completed in a single production run but are progressively fulfilled through multiple batches. This approach enhances production stability and facilitates more effective quality control. Furthermore, to address urgent requirements from international clients, some supply systems implement safety stock mechanisms. Safety stock provides a buffer during sudden order surges or logistical delays, ensuring clients' production schedules remain unaffected. For long-term collaborative projects, this inventory management approach significantly enhances supply reliability.

B2B Cooperation Process
Within the plant extract industry, most enterprises undergo a progression from technical evaluation to sustained collaboration when procuring raw materials. For the supply of Pomegranate Extract Punicalagin In Stock, we have established a B2B procurement cooperation process tailored for international clients, enabling a seamless transition from small-scale trials to long-term, stable supply.
Our Service Processes
Our free service hotline:88346301
Pre-sales Consultancy
1
>>
Confirmation of order
2
>>
Production
3
>>
Multi-Channel Shipping
4
>>
Confirmation of receipt
5
>>
After-sale Services
6
1. Cooperation typically commences with technical requirement confirmation. Clients define specifications based on product formulations and target market demands, such as Punicalagin content, polyphenol ratios, solvent residue standards, and microbiological parameters. As quality standards may vary across applications-e.g., dietary supplements versus cosmetic ingredients-detailed communication during initial stages is paramount.
2. This is followed by the sample validation phase. During this phase, clients typically conduct laboratory testing or small-scale formulation trials on samples to confirm stability and compatibility in practical applications. Sample batch data serves as a critical reference benchmark for future production batches, ensuring consistency in component content and quality metrics.
3. Upon sample validation completion, the partnership advances to supply planning. Here, both parties establish annual procurement plans, delivery cycles, and inventory strategies based on projected purchase volumes. For clients with long-term procurement requirements, a continuous supply mechanism is usually established to prevent raw material shortages caused by market demand fluctuations.
4. The final stage involves bulk production and delivery. Products are manufactured and quality-inspected according to the established production schedule, then transported via international logistics networks. All batches are accompanied by test reports and relevant documentation to facilitate smooth entry into international markets.
How Does the International Logistics System Operate?
As commodities in international trade, plant extracts typically require transportation across multiple countries, rendering the logistics system crucial within the supply chain. For the supply of Pomegranate Extract Punicalagin In Stock, we routinely employ established international shipping solutions to ensure products reach customers' locations safely and efficiently.
1. Regarding trade terms, common international trade conditions include FOB, CIF, CFR, DAP, and DDP. Different buyers select trade terms based on their import procedures. For instance, large raw material traders often prefer FOB or CIF, while brand companies may opt for DAP or DDP to reduce import operational complexity.
2. Transport methods are typically determined by order volume and urgency. Sea freight is the most common mode for tonnage-level orders due to its cost-effectiveness and suitability for bulk cargo. Sea freight transit times typically span several weeks, necessitating planning for production and shipment schedules. Air freight is primarily used for samples, urgent orders, or small-batch, high-value raw materials.

3. Logistics Services: Established stable partnerships with international logistics giants, including DHL, FEDEX, UPS, and TNT, to provide door-to-door delivery services.
Estimated Delivery Times:
Sample Orders: 3-5 working days
Small-batch stock orders (<100kg): 3-7 days
Bulk air freight orders: 5–8 days
Bulk sea freight orders: 15–30 days (depending on destination)
During cross-border shipments, preparing customs clearance documentation is equally critical. Commonly required documents include Certificate of Analysis (COA), Safety Data Sheet (SDS), product specifications, and Certificate of Origin. These documents facilitate importers' smooth completion of customs declarations and product regulatory compliance procedures.
What Does the Industrial Packaging System Include?
Plant extract powders are susceptible to moisture and oxygen exposure during transportation and storage, making a well-designed packaging system crucial for safeguarding product quality. For Pomegranate Extract Punicalagin In Stock, packaging typically employs a multi-layer structure to ensure product stability during long-distance transport.
Common packaging structures comprise two tiers: inner and outer packaging.
1. Inner packaging typically employs food-grade aluminium foil pouches or PE inner bags, materials offering excellent moisture and oxidation resistance to minimise air and humidity impact.
2. Outer packaging predominantly utilises cardboard drums or fibre drums, structures providing robust crush resistance suitable for extended transit and storage. Within international plant extract trade, 25kg/drum represents a prevalent standard packaging specification. This specification facilitates manual handling while accommodating palletised transport and container loading. For major clients, bespoke packaging or palletised shipping solutions can be arranged upon request.

Furthermore, given significant temperature and humidity fluctuations during maritime transit, packaging designs often incorporate moisture-proofing measures such as sealed structures or desiccants. These precautions effectively mitigate risks of moisture ingress or oxidation during transit, thereby ensuring consistent product quality upon arrival.
What Are the Synergistic Components and Product Matrix?
Punicalagin stands as one of the core high-value ingredients within our pomegranate extract product line. To support your development of more innovative and competitive end products, we recommend concurrently considering the following products exhibiting synergistic effects or complementary properties:
| Product Category | Key Ingredients | Primary Applications |
| Pomegranate Peel Polyphenols | Punicalagin, Ellagic Acid Complex | Broad-spectrum antioxidant, anti-inflammatory, and gut health |
| Ellagic Acid | 40%-98% | High-purity brightening and antioxidant raw material, functional skincare |
| Neohesperidin Dihydrochalcone | NHDC 98% | Bitterness masking, flavour modification; synergises with punicalagin to enhance palatability |
| Urolithin A | 98% | Mitochondrial health, muscle function support |
Access our comprehensive botanical extract product catalogue for further specialised ingredient solutions.(www.appchem.cn)
Reference
[1]WANGJ, SUN M, YU J, et al. Pomegranate seeds: a comprehensive review of traditional uses, chemical composition, and pharmacological properties[J]. Front Pharmacol, 2024; 15: 1401826. DOI: 10.3389/fphar.2024.1401826
[2]AVIRAM M, ROSENBLAT M. Pomegranate for your cardiovascular health[J]. Rambam Maimonides Med J, 2013; 4(2): e0013. DOI: 10.5041/RMMJ.10113
[3]DANESI F, FERGUSON L R. Could Pomegranate juice help in the control of inflammatory diseases?[J]. Nutrients, 2017; 9(9): 958. DOI: 10.3390/nu9090958
[4]GHAEMI F, EMADZADEH M, ATKIN S L, et al. Impact of pomegranate juice on blood pressure: A systematic review and meta-analysis[J]. Phytother Res, 2023; 37(10): 4429-4441. DOI: 10.1002/ptr.7952







