NMN stands for β-nicotinamide mononucleotide and is a nucleotide derived from ribose and nicotinamide. Like nicotinamide ribose, NMN belongs to the B group of vitamins.
NMN is an important nicotinamide adenine dinucleotide (NAD+) supplement. In the human body, NMN can be converted to NAD+. NMN has anti-aging effect by increasing NAD+ level. At present, three countries have conducted clinical studies on it: China, the United States and Japan. As of September 2022, a total of 38 international NMN-related human clinical trials have been registered, including 20 in Japan, 12 in the United States, and 6 in China, and 11 clinical trial results have been published.
01 Current market and regulations in China
NMN entered the Chinese market around 2018 and now has a large market share. Iresearch data show that in 2020, China's NMN ingredient health care products market size reached 5.106 billion yuan, a year-on-year increase of 34.87%, is expected to climb to 27.013 billion yuan by 2023 with a growth rate of nearly 70.25%, the industry development speed is fast. At the same time, consumers have a certain degree of awareness of NMN ingredients. Data show that more than 60% of Chinese consumers have had some understanding of NMN in 2021, of which 24.6% have specialized understanding of NMN products. At the same time, Chinese consumers' cognition of the functions of NMN products is mainly to enhance immunity, improve sleep and delay aging.
NMN has not yet obtained the license of new food raw materials, food additives, health food and drugs in China. Consumers can purchase NMN products through cross-border e-commerce. NMN products sold on China's cross-border e-commerce mainly come from the United States, Canada, Japan, Germany, Australia, Switzerland, Hong Kong and other countries and regions.
02 Market and regulatory status in Japan
Japan is the first country in the world to approve NMN as a food raw material. In July 2020, the Ministry of Health and Welfare of Japan added NMN to the list of health food raw materials - Ingredients not considered as drugs.

Japan is also the first country to conduct research on NMN. In 2005, Shinichiro Imai, a Japanese professor at the University of Washington, started NMN research. Subsequently, Japan Oriental Yeast Co., Ltd. sought cooperation with Professor Imai and began industrial development and mass production of NMN. In 2013, Imai found that when NMN was given to mice, there was an overall improvement in aging: hair became thickened and shiny, the average life span increased from two months (equivalent to six human years) to 4.6 months, and life expectancy increased 2.3-fold. In January 2020, NMN was confirmed to be safe for human use for the first time in the world.
In 2015, Japan's NHK program NEXTWORLD: Our Future featured NMN at large length, and NMN caused a national storm in Japan. NMN continues to be popular in the Japanese market, with the NMN Japanese market worth about 2.6 billion yen in 2020 and expected to reach 14.8 billion yen by 2027, according to Japanese research agency Fuji Economics.
At present, the main forms of NMN supplements in the Japanese market include capsules, chewable tablets, powder forms and liquid forms. NMN supplements in Japan will especially emphasize that the raw materials of NMN come from Japan, and use a variety of functional ingredients together to increase health effects.
Toyama-ya NMN supplement, which uses native Japanese NMN raw materials and combines them with red grape leaf extract, astaxanthin, salmon nasal cartilage extract, vitamin B2, and biotin.
03 Market and regulatory status in the United States
NMN has had a bumpy road to compliance in the US. Since 2020, a total of four companies have applied for NMN as a new dietary ingredient, but the FDA objected to the NDI notifications submitted by several companies on the grounds that "there is not enough evidence to prove that NMN supplementation is safe". By May 16 last year, SyncoZymes had received an official response from the FDA that β-NMN, produced by Syncozymes, had received NDI approval for use in dietary supplements. However, on November 11, the same year, the FDA responded to a new dietary ingredient (NDI) notice from Inner Mongolia Jindawei Company, and pointed out that β-NMN could not be sold as a dietary supplement because it had been studied as a new drug. At present, NMN dietary supplement manufacturers and associations are actively appealing to the FDA, hoping to exercise enforcement discretion against NMN. For now, however, the FDA does not appear to be telling retailers to stop selling NMN products.
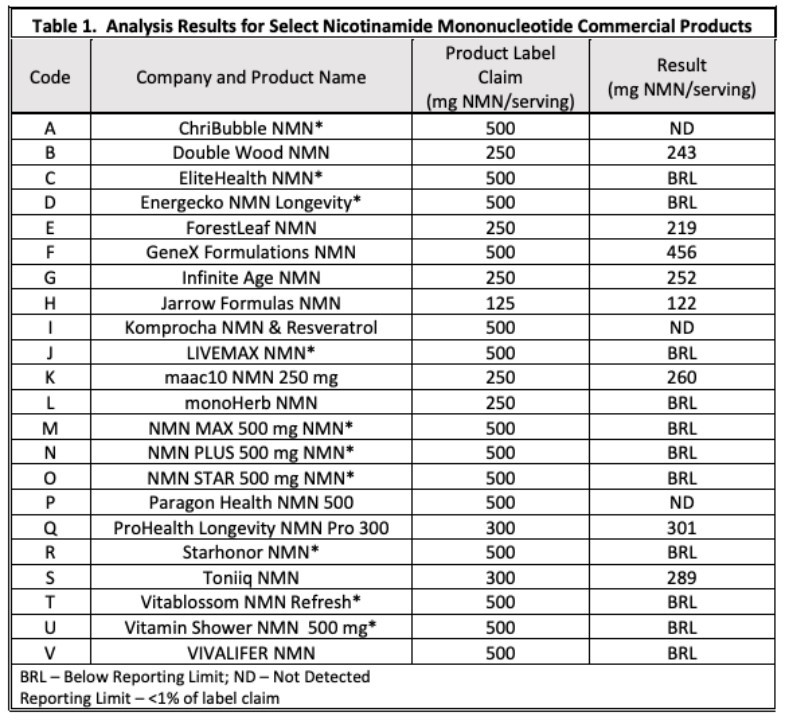
In the context of increasing global population aging, the market attention and scale of NMN are bound to continue to increase. According to estimates, the current domestic NMN market space corresponding to 1% of the population of health care products is 30.4 billion yuan, and the long-term market of the industry is expected to reach 100 billion yuan in the future.
